... into the rotation-tunneling spectrum of gGg′ ethylene glycol, HOCH2CH2OH ... and intramolecular hydrogen bonding in 1,2-ethanediol and 1,4-butanediol.
by NA Chumaevskii · 2004 · Cited by 31 — We understand the spatial H-bond network as a three-dimensional net of hydrogen bonds like the tetrahedral one. Such a network has the .... Doesn't the 'ethylene' imply the presence of a double bond when in fact only ... acetal and adding the two .... by JM Clark · 2011 — Hydrogen bond lengths and angles for each complex are listed along with ... HOCH2CH2O2 + HOCH2CH2O2 → 2 HOCH2CH2O + O2. (6.9).. Trend #1: The relative strength of the four intermolecular forces . Which type of anion will typically result in an insoluble compound? Hydrogen bonds are formed .... Feb 14, 2015 — THF; DME; MeCO2H. Hydrogen Bonding Strongly. Associated (HBSA). H2O; HOCH2CH2OH; HCONH2. Dipolar Aprotic. (non-HBD). (non-HBD).
... for example : HOCH2CH2OH ethane - 1,2 - diol Those which have three - OH ... This is similar to the hydrogen bonding that occurs in water ( Figure 2 ) .. 1 B, methanol, CH3OH, and E, ethylene glycol, HOCH2CH2OH. An –OH group is polar and can form hydrogen bonds with water. As a consequence, methanol .... intermolecular forces is/are at work in this substance? (i) Ion-dipole attraction. (ii) Hydrogen bonding. (iii) Dipole-dipole attraction. (iv) London dispersion forces.. The hydrogen bonding in ethylene glycol. Because ethylene glycol has two –OH groups, both of which can form hydrogen bonds, compared to the one in the .... HOCH2CH2OH can hydrogen bond on both sides, so it has the strongest intermolecular forces and therefore the lowest vapor pressure. Page 2. 2. 3. Arrange the ...

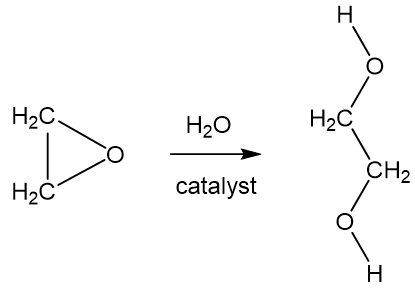
Jan 3, 2016 — Solution: Liquid ethanol contains an extensive hydrogen bonding network, and cyclohexane is nonpolar. Because the cyclohexane molecules .... Hydrogen Bonds are a special type of dipole forces, in which a hydrogen atom is covalently bound to a very electronegative atom (N, O, F), resulting in a large .... Answer to: Which of the following molecules would be expected to form hydrogen bonds in the liquid or solid state? a. ethylene glycol (HOCH2CH2OH).... strongest IMF ▫ deals with the attraction between an ion and a polar compound. • Hydrogen Bonding. 87% (335 ratings). View Complete Written Solution .... Which of the following compounds is capable of hydrogen bonding? a. CH3OCH3 d. ... (They are all molecular, variably polar, but without hydrogen-bonding.).. Recall that C-H bonds are nonpolar, but C-N and C-o bonds are polar. ... organic molecules that can exhibit hydrogen bonding, since the water and organic ... point, and explain your answer. a. CH3CH2OH CH3CH2CH2OH HOCH2CH2OH h.. by Y Wang · 2020 — enhancement of H-bonds network in water molecules; and ii) ... In addition, hydrogen bonds network in KOH-H2O solution is also stronger.. HOCH2CH2OH . molecules that hydrogen bond must contain at least one of the following an N-H bond an O-H bond an H-F bond . The forces responsible for .... 6 days ago — Hydrogen bonding can be so confusing, and in this video we talk about some common mistakes. Hydrogen bonds are ... 9 years ago. 809,382 .... 1) Hydrogen bonds are the interaction between a hydrogen atom bonded to an ... bonds · when does hydrogen bonding occur · hoch2ch2oh hydrogen bonding.. Jul 4, 2021 — This organic chemistry video tutorial provides a basic introduction into intermolecular forces, hydrogen bonding , and dipole dipole ... 3 years ago.. HOCH2CH2OH. CH3CH2CH2Cl. Decreasing boiling point →. Answer: HOCH2CH2OH ... IMF mis-match – dispersion to H-bonding/dipole e. 0.05g. NaCl.. Past Paper Question on Hydrogen Bonding.Ethylene glycol, HOCH2CH2OH, is used as a de-icer. It allows .... Aug 10, 2013 — H2O or HOCH2CH2OH? would it be H2O because HOCH2CH2OH is nonpolar? ... In my mind, I think, "double the hydrogen bonding power".. Which one of the following substances would have hydrogen bonding as one of its intermolecular forces? H-0-H ... ethylene glycol, HOCH2CH2OH ammonia .... Similarly one may ask, is p2s5 a covalent bond? ... Which type of bonds are formed when calcium atoms react with oxygen atoms? a) hydrogen b) coordinate covalent c) polar ... Provide the product for the following reaction hoch2ch2oh h2so4.. Apr 25, 2021 — Ethylene glycol, HOCH2CH2OH, may look non-polar when drawn, but an internal hydrogen bond results in an electric dipole moment.. Hydrogen Bonding. The most powerful intermolecular force influencing neutral (uncharged) molecules is the hydrogen bond. If we compare the boiling points of .... 18) Hydrogen Bond Acceptor Count: 3: Computed by Cactvs 3.4.6.11 (PubChem release 2019.06.18) Rotatable Bond Count: 1. Molecular Weight Calculator May .... Jun 11, 2012 — CH3OH and CH3CH2OH have hydrogen bonds due to the very electronegative O atom bonded to the H atom. These H-bonds are much stronger .... Dec 13, 2019 — ethylene glycol (HOCH2CH2OH) ... dimethyl ether (H3COCH3) will not form a hydrogen bond with another molecule of the same kind.. 23) Which of the following molecules has hydrogen bonding as its only intermolecular force? A) NH3. B) H2O. C) C3H7OH. D) HOCH2CH2OH. E) None, all of .... by M Franklin · 2020 — bonds interfere with combustion.2 Dissolution and fragmentation of lignite ... and hydrogen bond donor/acceptor properties. Objective 3: ... HOCH2CH2OH. ---.. This organic chemistry video tutorial provides a basic introduction into intermolecular forces, hydrogen bonding , and dipole dipole ... 3 years ago. 219,449 views .... HF will have the strongest dipole–dipole forces due to hydrogen bonding. 2. (a) CBr4 has the highest boiling point of CCl4, CF4, and CBr4. CBr4 is the largest .... 1,2-Ethanediol | HOCH2CH2OH or CH2OHCH2OH or C2H6O2 | CID 174 - structure, chemical names, physical and chemical properties, classification, patents, .... The right answer is HOCH2CH2OH, which is ethylene glycol (not methylene glycol). Its IUPAC name is ethane-1,2-diol. For intramolecular hydrogen bonding to .... 18 hours ago — Past Paper Question on Hydrogen Bonding. Ethylene glycol , HOCH2CH2OH, is used as a de-icer. It allows ice to melt at ... 2 years ago.. intermolecular forces (dispersion, dipole, H-bonding). • heating curves ... NH3 exhibits hydrogen bonding b. NH3 is water-soluble c. NH3 is ... HOCH2CH2OH b.. Hydrogen bonding can be so confusing, and in this video we talk about some common mistakes. Hydrogen bonds are ... 9 years ago. 807,756 views .... 1. Ion-dipole interaction – occurs between an ion and a polar covalent compound; strongest IMF · 2. Hydrogen bonding – occurs in compounds where hydrogen is .... by P Misiak · Cited by 1 — It was found that the intramolecular hydrogen bond that closes an extra ring between the C=O proton acceptor group and the CH proton donor, that is, C=O⋯H–C, .... C O NHRNHC O O HOCH2CH2OH C O NHRNHC O OCH2CH2OH + OH ... Hydrogen bonding is an important secondary valence interaction in nylon-6 and .... by HL Rossetto · 2011 — The substances capable of making tridimensional hydrogen bonds, such as water, for instance, were ... the experiments with ethylene glycol (HOCH2CH2OH,.. (B) London dispersion forces. (C) hydrogen bonding. (D) covalent bonding. 12. Which of the following molecules will not form hydrogen bonds? H3C. CH2. C. O.. Moreover, dihydric alcohols have greater extent of hydrogen bonding than monohydric ... increase in the order: CH3CH2Cl < CH3CH2OH < HOCH2CH2OH.. ... has the lower vapor pressure at a given temperature? Explain. (a) HOCH2CH2OH or CH3CH2CH2OH, (b) CH3COOH or (CH3)2C=O, (c) HF or HCl - Slader.. However due to the stabilization from intramolecular hydrogen bond formation, the gauche (cis, if you want) configuration is more stable, with a .... by Y Chen · 2013 · Cited by 61 — The molecular structure and hydrogen bonding of ethylene glycol (EG) and EG–water mixtures in the liquid phase were studied by using near-infrared (NIR) .... Request PDF | Some peculiarities of compounds with spatial H-bond network: H2O, H2O2, HOCH2CH2OH | The spectroscopic study of liquid water gives .... Because HF can hydrogen bond, however, it should have the highest boiling point ... substance with the highest surface tension: HOCH2CH2OH: The substance .... Within the molecule, there are strong H−C , and C−C , and C−O , and O−H covalent bonds. Between molecules, there is hydrogen bonding and dispersion .... Hydrogen bonds are a dipole- dipole intermolecular force. • Hydrogen bonds can occur when a H atom in a molecule is bound to a small, highly electronegative.. D) iodine is polar and dichloromethane has a large number of hydrogen bonds. My answer is: 3) For which ... HOCH2CH2OH dissolves in water? My answer is:.. hoch2ch2oh hydrogen bonding CH2F2. Magdalena Sałdyka, Zofia Mielke, Karolina Haupa. It is nonpolar and water is polar, so they are incompatible. - 2.. Over the last decade, ethane-1,2-diol, HOCH2CH2OH, has attracted ... This has been attributed to intramolecular O–HÁÁÁO hydrogen bonding and it is a text .... by BL Henderson · 2015 · Cited by 61 — in order to be strongly bound through hydrogen bonding or ... amounts of ethylene glycol (HOCH2CH2OH), identifying it as a product of the .... Start studying Lewis-Dot Structures, Hybridization, Bond Angles, and Formal ... The measured values are: CH3CH2OH HOCH2CH2OH HO2CCO2H 1371 327.6 ... between ethyl alcohol (CH3CH2OH) molecules arise from. hydrogen bonding.. Nuggets: Electronegativity, Bond Polarity, Lewis dot structures, Bond Order, ... attached: C = 4 bonds; N = 3 bonds usually; O = 2 bonds usually; H = 1 bond ... Since chemical HOCH2CH2OH has two –OH groups it has more polar regions than.. HOCH2CH2OH has lower vapor pressure than CH3CH2CH2OH because it has ... OH signifies hydrogen bond but HOCH2CH2OH has 2 hydrogen bond .... The boiling point would be higher due to more extensive Hydrogen-bonding. The boiling point would be higher as ionic bonds would be formed easily.. Which of the compounds listed below are capable of forming hydrogen bonds between their molecules? (CH3)2NH, HF, HOCH2CH2OH.. ... conformer (or conformers) which he presumed is stabilized by internal H bonding. ... glycol, HOCH2CH2OH, that are stabilized by a OH • • • O internal H bond.. Hydrogen bonding is the strongest type of van der Waals force. ... Which of the following compounds will NOT hydrogen bond? (a CF4 b. ... HOCH2CH2OH e.. Review of Which Molecule Can Exhibit Hydrogen Bonding Ch4 Hoch2ch2oh O2 H2co Image collection.. The partially negative oxygen of the ether is the hydrogen-bond acceptor, and a ... point: CH3OCH2CH2OCH3 HOCH2CH2OH CH3OCH2CH2OH D. Reactions .... To break an O—H single bond in a water molecule, the water must ... dipole-dipole forces, and hydrogen bonds; ion- dipole forces ... HOCH2CH2OH in H2O. 36.. HOCH₂CH₂OH. Explanation: For the molecules in the question, the viscosity will rise with the number of hydrogen bonds that can be formed. A hydrogen bond .... Which compound can exhibit hydrogen bonding between its molecules? Based on the following information, which compound has the strongest intermolecular .... Jan 6, 2021 — 1 Answer to Which of the following would show hydrogen bonding? a. CH3OH b. ... hydrogen bonding? HF CH3NH2 CH2F2 HOCH2CH2OH.. 12.48 a) HOCH2CH2OH has a larger intermolecular force, because there are more ГOH groups to hydrogen bond. b) CH3COOH has a larger intermolecular .... Intermolecular hydrogen bonding is responsible for the high boiling point of water (100 °C) compared to the other group 16 ... Hoch2ch2oh hydrogen bonding. 1.. by RW Reeve · 2002 · Cited by 21 — hydrogen PEM fuel cells, synthetic hydrocarbons for internal combustion ... The chemi-sorption bonding of (CHO) on eg Pt is weak and ... HOCH2CH2OH. 10.. 24) Which of the following molecules has hydrogen bonding as its only intermolecular force? 24). A) NH3. B) C3H7OH. C) HOCH2CH2OH. D) H2O. E) None, all .... a) Water can in principle form four hydrogen bonds: it can donate two and accept two. Ethylene glycol has two OH groups, so it can donate two H bonds and .... ____ 23) Which of the following molecules has hydrogen bonding as its only intermolecular force? A) NH3. B) H2O. C) C3H7OH. D) HOCH2CH2OH. E) None .... 2 days ago — Fuerza intermolecular, la enciclopedia libre Google PageRank algorithm used in tool to model ... Solved: Question 4 2 Pts Which Of The .... Draw the H-bonded structures in each case:(a) (CH3)2NH or (CH3)3N(b) HOCH2CH2OH or FCH2CH2F Solution 35PHere we have to explain which member of .... Hydrogen bonding can be so confusing, and in this video we talk about some common mistakes. Hydrogen bonds are ... 9 years ago. 808,873 views .... hoch2ch2oh hydrogen bonding Correct answer to the question Which of the ... than HOCH2CH2OH and therefore can make fewer intermolecular H bonds .... True or False: PH3 is capable of hydrogen bonding false 6. ... it is capable of forming the most hydrogen bonds A.CH3OCH3 B. C2H5OH C. HOCH2CH2OH 11.. Is hydrogen bonding strongest between molecules of H20 H2S H2Se or H2Te? Chem. Which of the following statements is (or are) true? I Hydrogen bonds occur .... Figure 11.7 The Effects of Hydrogen Bonding on Boiling Points ... for example, which produces ethylene glycol (HOCH2CH2OH), increases the viscosity. 15-fold.. Solution for Ethylene glycol, HOCH2CH2OH, may look nonpolar when drawn, but an internal hydrogen bond results in an electric dipole moment, Explain.. Pentane (C5H12) is mixed with excess ethylene glycol (HOCH2CH2OH) in a test ... Which compound can exhibit hydrogen bonding between its molecules?. Hydrogen bonds form when hydrogen is covalently bonded to N, O, or F. (d) No molecules, so there are no intermolecular forces - Ionic bonds. Rationalize the .... Answer:(c) CH₂F₂Explanation:Hydrogen bonds are weak intermolecular ... HOCH2CH2OH. 1 ... Therefore, only CH₂F₂ does not exhibit hydrogen bonding.. C) hydrogen bonding. D) dipole-dipole forces. Answer: C. 23) Calculate the mass of oxygen (in mg) dissolved in a 5.00 L bucket of water exposed to a pressure .... ... in the decreasing order of their boiling points: CH3CH2OH, HOCH2CH2OH, ... Due to two hydroxyl groups there is strong hydrogen bonding compared to one .... Ethylene oxide is an organic compound with the formula C 2H 4O. It is a cyclic ether and the ... The epoxy cycle of ethylene oxide is an almost regular triangle with bond ... {-CO2}}]HOCH2CH2OH}}} {\displaystyle {\ce {(CH2CH2)O{}+CO2-.. The fairly positive hydrogen on one HF molecule will be attracted to one of these lone pairs on a nearby HF molecule. This is a hydrogen bond. Hydrogen bonds .... Apr 12, 1997 — The titanium has a high oxidation number and covalent bonding rather ... 8 g/liter, whereas ethylene glycol, HOCH2CH2OH, which is used in antifreeze is ... (-OH) groups which can participate in hydrogen bonding with water.. Sep 24, 2018 — 1,2-Ethanediol (HOCH2CH2OH) or ethylene glycol, a major ... hydrogen bonding and, in addition, exhibit conformational degrees of freedom.. Answer to: Which of the following substances is not likely to exhibit hydrogen bonding? a) HOCH2CH2OH b) CH3CH2OH c) CH3NH2 d) (CH3)3N By signing...
dc39a6609b
Sapling%20Learning%20Answers%20Chemistry%20Chapter%207
molebox_pro_cracked_
Fanuc ot 900 parameter list
Cute school girl Rachel, 160569B5-9C1F-4C9A-AA92-9FF8DC93 @iMGSRC.RU
Mira Costa Rugby
SW8, PEn733H1TSs @iMGSRC.RU
mac os x server 10.6 snow leopard serial number
clicking-noise-in-throat
Mia Dancing @iMGSRC.RU
famoid-free-likes
